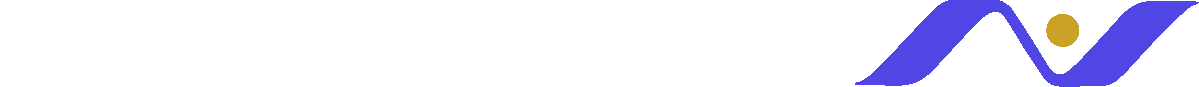The latest news from Neuromics, customers, collaborators, and friends. Here we will post publications, data, new reagents, and methods.
For additional information on Neuroscience updates check out our Neuroscience Blog.
Human Primary Cells, iPSC Derived Cells & Cell Lines
More Fascinating Research with GFP-Expressing Cells
Over the past few years, researchers from Dartmouth have utilized our GFP-expressing U87 MG cells (cat. TR01-GFP) in surgical imaging research. They have been using the cells to evaluate various contrast agents for fluorescent-guided surgery by implanting our cells in mouse models. Last week, they published another study with fascinating results.
In their initial paper, released last year, the researchers identified TMR-PEG1k as the preferred contrast agent when compared with others. Then, earlier this year, the same group released a study highlighting the need for standardized methods to study contrast agents.
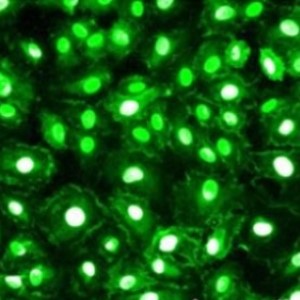
Image: Neuromics GFP Expressing U87 MG Cells were incubated with two potential contrast agents in surgical research.
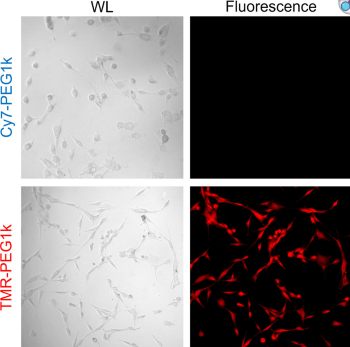
In this week’s research, the investigators developed a near-infrared version of TMR-PEG1k to address some of the agent's shortcomings. Being in the visible regime, TMR-PEG1k is excellent for superficial tissue, but doesn’t allow for subsurface penetration, unlike a near-infrared agent. Therefore, the researchers developed a cyanine 7 version of the agent as an alternative.
Primary Human Astrocyte and Human Umbilical Cell (HUVES) Organoids
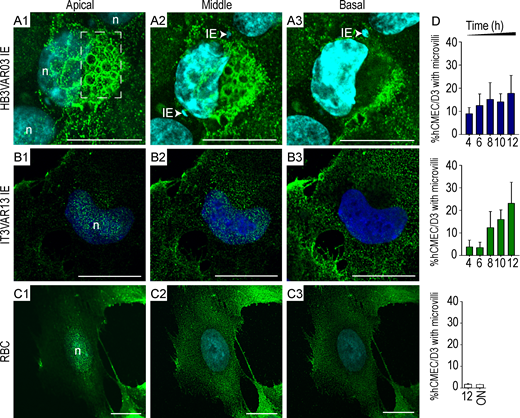
Yvonne Adams, Anja T.R. Jensen (University of Copenhagen) manufactured Organioids using Neuromics' Primary Human Astrocytes and Human Brain Microvascular Endothelial Cells to study Plasmodium falciparum erythrocyte membrane protein 1 variants induce cell swelling and disrupt the blood–brain barrier in cerebral malaria.
Cerebral malaria (CM) is caused by the binding of Plasmodium falciparum–infected erythrocytes (IEs) to the brain microvasculature, leading to inflammation, vessel occlusion, and cerebral swelling.
Image 1: 3D BBB spheroids are composed of three different cell types. (A) FACS histogram showing pericytes (NG2, neural/glial antigen 2), astrocytes (GFAP, glial fibrillary acidic protein), and human cerebral microvascular endothelial cells (hCMEC/D3 and CD31).
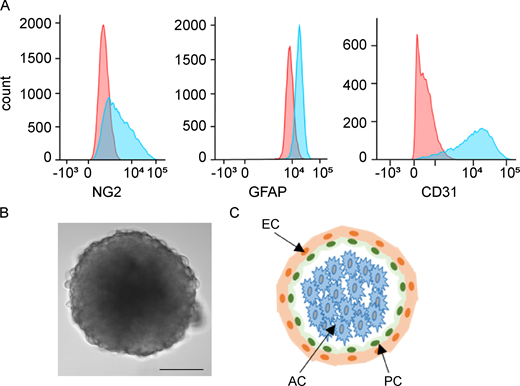
Image2: ICAM-1–enriched microvilli and transmigratory ring/docking structures on brain hCMEC/D3 endothelial cells. (A–C) hCMEC/D3 brain endothelial cells were incubated with parasites.
This is first time the presence of intact, mature P. falciparum IEs within brain microvascular endothelial cells both in vitro and in vivo. The data on the enhanced binding and internalization of IEs suggest that the same approach will be necessary to counteract not only the effects of cytoadhesion but also the subsequent contribution to potentially lethal brain swelling in CM.
Neuromics Expands Human iPSC Portfolio
As part of our commitment to supporting innovative research, Neuromics continually seeks to introduce new human cell types that meet the needs of researchers. Earlier this month, we introduced several new cell types for studying diabetes and obesity. Now, we've turned our attention back to our selection of human iPSC-related cells, with the addition of three cell types. As a reminder, we offer a wide range of iPSC-derived neural cell types (check them out).
iPSC-derived models are transforming neuroscience research - especially for diseases like ALS, Parkinson’s, and Alzheimer’s. Models from diseased donors open the door for new assays and drug discovery approaches.
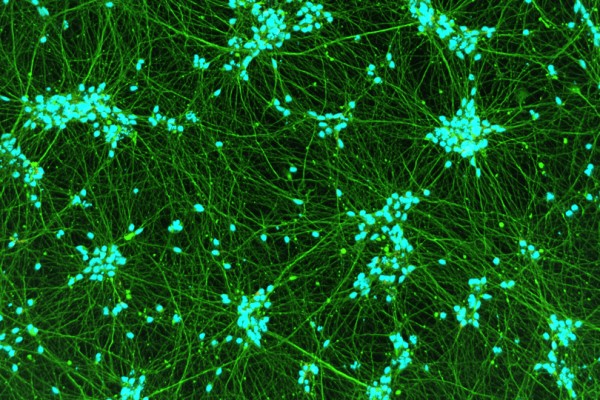
Image: Neuromics Human iPSC - Sensory Neurons (cat. IPS006) in culture.
Here are the new cell types:
- Human iPSC (cat. IPS013): a versatile cell line for custom differentiation or disease modeling.
- Human iPSC - Motor Neurons (ALS Patient, Sporadic) (cat. IPS014): derived from a patient with sporadic ALS, providing a physiologically relevant model for studying disease mechanisms
- Human iPSC - Motor Neurons (TDP-43 M337V, Homozygous) (cat. IPS015): a genetic model capturing a well-characterized ALS-linked mutation in TARDBP.
Studying the Tumor Microenvironment? You Need Our CAFs
With colorectal cancer (CRC) being one of the most prevalent cancers worldwide, understanding chemotherapy resistance in the CRC tumor microenvironment (TME) is an important goal. As we’ve seen in many other publications using our human cancer-associated fibroblasts (CAFs), CAFs play a huge role in the TME.
Our CAFs have been used in a handful of studies in 2D and 3D TME models, where they demonstrate their ability to complicate treatment objectives. Overall, the mechanisms by which CAFs contribute to chemotherapy resistance in the TME are poorly understood.
Image: DKK1 expression in Neuromics colorectal CAFs when subjected to chemotherapeutic agents.
Researchers from the University of Kentucky utilized our colorectal CAFs (cat. CAF115) in a new paper examining these mechanisms. In their cell models, they found that CAFs secrete DKK1, a protein associated with tumor growth and metastasis, when subjected to chemotherapy treatments for CRC. Their results identified CAF-secreted DKK1 as a major player in resistance to treatment. Furthermore, they recommend looking into treatments that target DKK1 to counteract chemotherapy resistance.
You can check out the full paper here.